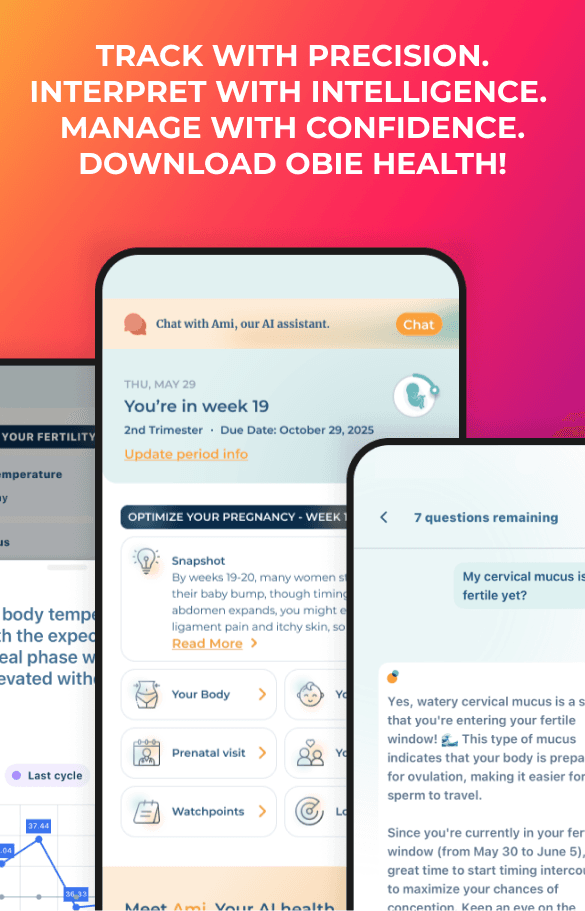
Polyhydramnios - Too Much Amniotic Fluid
Pregnancy
Obie Editorial Team
Polyhydramnios refers to the presence of an excessive amount of amniotic fluid in the uterus during pregnancy, which is an essential component surrounding your growing baby, playing a crucial role in their development. Generally, fetal urination and swallowing help maintain this fluid balance. If there is an increase, it might be due to disruptions in these processes. Amniotic fluid typically grows until around 34 weeks of gestation and minimally decreases as you approach your due date.
Determining Amniotic Fluid Quantity: Empower Your Knowledge
After the 20-week mark, healthcare providers typically assess amniotic fluid volume using either the deepest vertical pocket (DVP) method or the amniotic fluid index (AFI). In cases of multiple pregnancies, DVP is the preferred diagnostic tool.
Understanding the Deepest Vertical Pocket (DVP)
The DVP evaluates the deepest section of amniotic fluid in centimeters. Polyhydramnios is determined if the DVP reading is ≥ 8.0 cm. Identifying such metrics empowers you and your healthcare provider in decision-making based on precise data.
The Amniotic Fluid Index (AFI) as a Diagnostic Tool
The Amniotic Fluid Index (AFI) is a standard method for measuring amniotic fluid levels. Evaluating the AFI can be the first step your doctor takes in diagnosing polyhydramnios. At about 32 weeks, a typical AFI measurement ranges from 86 mm to 242 mm. On the spectrum, anything ≥ 24 cm or 25 cm usually indicates polyhydramnios.
Exploring Polyhydramnios: Mild, Moderate, and Severe
Polyhydramnios can be classified into mild, moderate, or severe, based on the AFI or DVP measurements. Mild cases, the most common (65-70%), have AFI of 24.0 to 29.9 cm or DVP of 8 to 11 cm. Moderate is marked by AFI of 30.0 to 34.9 cm or DVP of 12 to 15 cm. Severe cases (less than 15%), with AFI ≥ 35 cm or DVP ≥ 16 cm, require immediate attention.
Identifying Causes: Understanding the Underlying Factors
When polyhydramnios is detected, additional tests are vital to uncover possible causes. Often, no specific reason (idiopathic polyhydramnios) is identified, accounting for over 60% of cases. Fetal issues such as neural tube defects or abdominal wall defects account for 20%, while maternal conditions like diabetes or Rh disease influence the other 20%.
- More than 60% are idiopathic, with no identifiable cause.
- 20% are attributed to fetal complications, such as neural tube defects.
- 20% involve maternal factors, like diabetes or Rh disease.
Understanding how altered fetal swallowing or overproduction of urine can affect fluid levels helps in recognizing conditions that need intervention. Be aware that any serious anomaly causing nonimmune hydrops fetalis (NIHF) might also lead to polyhydramnios. In maternal diabetes, for instance, maternal hyperglycemia can result in fetal hyperglycemia and excess diuresis.
Evaluation after Detecting Polyhydramnios: Taking Charge
Recognizing idiopathic polyhydramnios often involves ruling out other causes through targeted ultrasonography to examine fetal structure. It's crucial to assess fetal growth since idiopathic polyhydramnios may accompany macrosomia. Even when ultrasound does not reveal anomalies, ongoing monitoring is recommended.
- Screen for diabetes and infections, such as syphilis, in pregnancies with mild polyhydramnios.
- In cases of severe polyhydramnios presenting early, a detailed ultrasound and medical history review are warranted.
Management: Strategies for a Healthy Pregnancy
Typically, mild polyhydramnios seen later in pregnancy doesn't need treatment. For severe cases causing symptoms like maternal discomfort, amnioreduction might provide relief, though the fluid commonly resurfaces. Use of Indomethacin to manage fluid isn’t recommended due to potential neonatal complications and lack of demonstrated benefits.
Antepartum Care and Monitoring
With polyhydramnios, meticulous monitoring of fetal development is crucial. Although idiopathic forms don't usually lead to preterm birth, severe cases are linked to increased risks. Tracking the pregnancy progression helps identify potential underlying issues.
Continuing ultrasounds to monitor fluid levels or fetal growth might be warranted. Nothing in current guidelines specifically mandates increased monitoring for isolated polyhydramnios, but some cases benefit from proactive follow-up.
Delivering with Confidence: Intrapartum Management
During labor, if polyhydramnios is present, there’s an increased chance of nonvertex presentations and potential challenges with labor progress. By determining fetal position and opting for potential manual correction methods, delivery plans can be adjusted. Be aware that cesarean deliveries are more frequent with polyhydramnios, and constant monitoring ensures maternal and fetal safety.
Newborn Care Planning
Guiding you through delivery, we also prepare for your newborn's needs, particularly if previous assessments suggest any congenital issues. Having pediatric support readily available ensures that any necessary care can be delivered promptly.
Real empowerment comes from understanding your situation and knowing how you and your care team can work together to navigate your pregnancy journey confidently. By staying informed and engaged at every step, you make choices that best support your and your baby’s health.